
So technically, both atomic mass and average atomic mass are atomic masses, but one represents a single atom, and the other represents the average of the isotopes. It is the abundance of isotopes of an element found naturally, expressed in percentages. Atomic Mass (u) Nuclear Mass(GeV/c2: Binding Energy(MeV) Spin: Natural Abund. Element: Nitrogen Symbol: N Atomic Mass: 14.0067 of Atoms: 2. Its unit is also amu.īut the average atomic mass depends on one more critical aspect, the isotopic abundance. Symbol: Ca Atomic Mass: 40.078 of Atoms: 3 Mass Percent: 81.104. mass 23 has been absorbed by the remainder of the element in such a manner as. The average atomic mass expresses the atomic mass of elements with isotopes. ( 156 ) 75 ( As ) 32 ( Na ) + 40 ( Ca ) + 12 ( C ) ( 157 ). 1 Da is defined as 1 12 of the mass of a free carbon-12 atom at rest in its ground state. Isotopes became the reason for calculating the average atomic masses, as we must consider an element's isotopes. The atomic mass (m a or m) is the mass of an atom.Although the SI unit of mass is the kilogram (symbol: kg), atomic mass is often expressed in the non-SI unit dalton (symbol: Da) equivalently, unified atomic mass unit (u). Then came isotopes, the atoms that differ slightly in atomic masses due to the varying number of neutrons in their nucleus. The unit of atomic mass is non-SI, amu (atomic mass unit). This calculation gives us the mass of a single atom of an element. But we can figure it out by adding up the number of protons and neutrons in the nucleus of an atom. Name of the isotope: Calcium-40 Ca-40 Symbol: 40 Ca or 4020 Ca Mass number A: 40 ( number of nucleons) Atomic number Z: 20 ( number of protons) Neutrons N: 20 Isotopic mass: 39.9625909 (2) u ( atomic weight of Calcium-40) Nuclide mass: 39.9516195 u (calculated nuclear mass without electrons) Mass excess: -34.84635 MeV Mass defect: 0.36720806. Element: Carbon Symbol: C Atomic Mass: 12.0107. Symbol: Ca Atomic Mass: 40 Classification: alkaline earth metals Protons: 20 Electrons: 20 Neutrons: 20 Color: silvery Discovery in: 1808 Density: 1.55 g/cm 3. Name Symbol Atomic weight Annotations 1 Hydrogen H 1.0079 g m 2 Helium He 4.0026 3 Lithium Li 6. 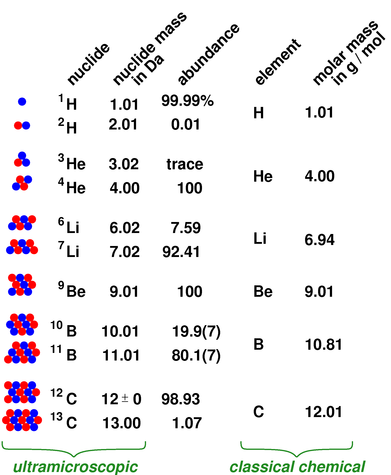 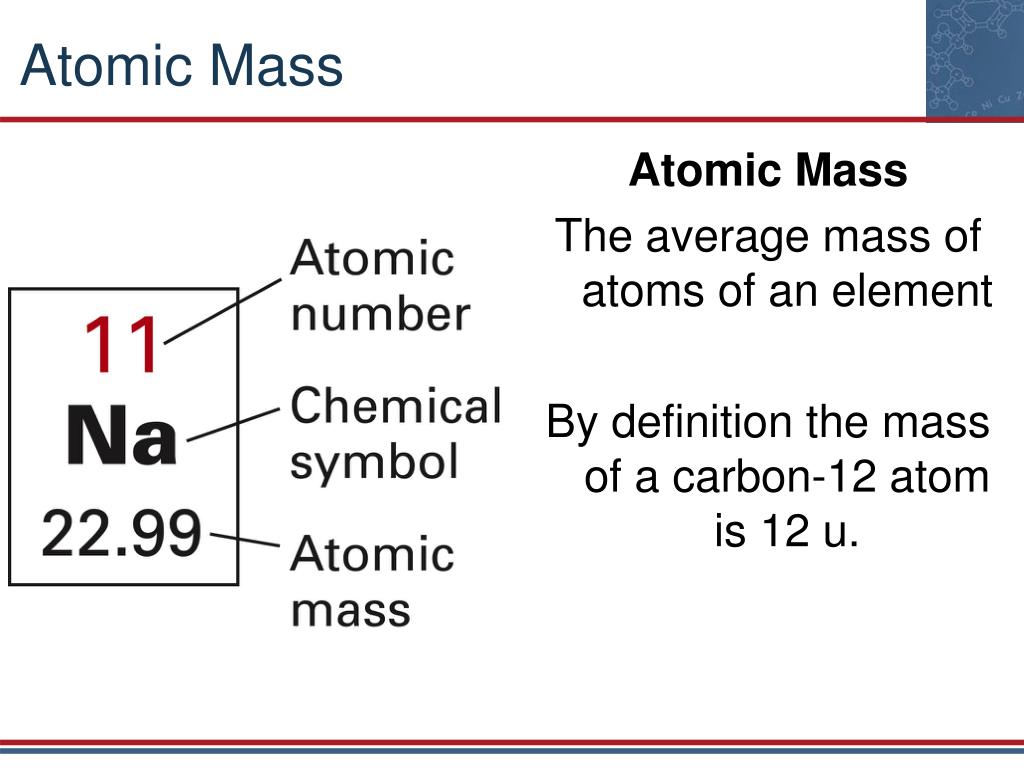
The isotope-amount ratio n( 44 Ca)/n( 40 Ca) ratio of calcium carbonate may serve as a paleothermometer to determine seawater temperatures in the past, making use of the temperature-dependent isotopic fractionation between 40 Ca. Scaled to the relative atomic mass, At(12C)12. The isotope-amount ratio n(44 Ca)/n(40 Ca) has been used to trace sources of calcium in soil and river water 184. It is the mass of a single atom of that element.Įxperimentally it is calculated by mass spectrometry (an analytical technique used to measure the mass-to-charge ratio of ions). Element: Calcium Symbol: Ca Atomic Mass: 40.078 of Atoms: 1 Mass Percent: 40.043. 1991 Table of standard atomic weights abridged to five significant figures. We have been familiar with the atomic mass of an element since we started learning about elements and their atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
